Bismuth-209
Q. How long will it take for three fourths of a sample of iodine-131 to decay?
16.2 days
Table of Contents
- Q. How long will it take for three fourths of a sample of iodine-131 to decay?
- Q. How much of a sample of radioisotope remains after one half life?
- Q. Which radioisotope decays the fastest?
- Q. Is Darmstadtium radioactive?
- Q. Is a short or long half-life more dangerous?
- Q. Is short half life dangerous?
- Q. Why do medical tracers have a short half life?
- Q. What does a short half life mean radiation?
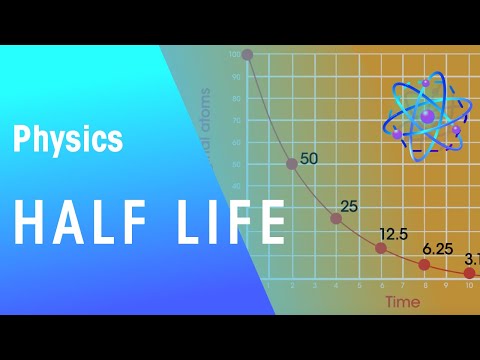
Q. How much of a sample of radioisotope remains after one half life?
After each half-life, half of the existing radioactive atoms have decayed into atoms of a new element. What are two ways that transmutation can occur?
Q. Which radioisotope decays the fastest?
37Ca
Q. Is Darmstadtium radioactive?
Darmstadtium is a radioactive, synthetic element about which little is known.
Q. Is a short or long half-life more dangerous?
Istopes with shorter half-lives are more intense. In nuclear waste, isotopes with very short half-lives, say a few days or even a few weeks, are not the major concern. They will decay to negligible amounts within a year or two. Isotopes with very long half-lives, more than 1000 years, are likely to be less intense.
Q. Is short half life dangerous?
Radioisotopes with short half-lives are dangerous for the straightforward reason that they can dose you very heavily (and fatally) in a short time. Such isotopes have been the main causes of radiation poisoning and death after above-ground explosions of nuclear weapons. Long-term isotopes are more complicated.
Q. Why do medical tracers have a short half life?
Different ligands form coordination complexes which give the technetium enhanced affinity for particular sites in the human body. Tc decays by gamma emission, with a half-life: 6.01 hours. The short half-life ensures that the body-concentration of the radioisotope falls effectively to zero in a few days.
Q. What does a short half life mean radiation?
However, even tiny pieces of material contain very many atoms . Some of its unstable nuclei decay in a short time, while others decay much later. The half-life of a radioactive isotope is the time taken for half the unstable nuclei in a sample to decay.